43 ingrezza isi
Neurocrine Biosciences Inc (NBIX) Q3 2020 Earnings Call Transcript I'll keep my comments brief and not reiterate all the financial information included in our earnings release to allow more time for Q&A. INGREZZA sales in the third quarter were $254 million and... Snippet Roundup: Firsts For Chinese CAR Companies ... - SeekingAlpha Xenazine is a benchmark owing to pricing considerations - Leerink analysts reckon on Austedo costing just $45,000 per patient per year versus Xenazine's $83,000 - and Austedo and Ingrezza's...
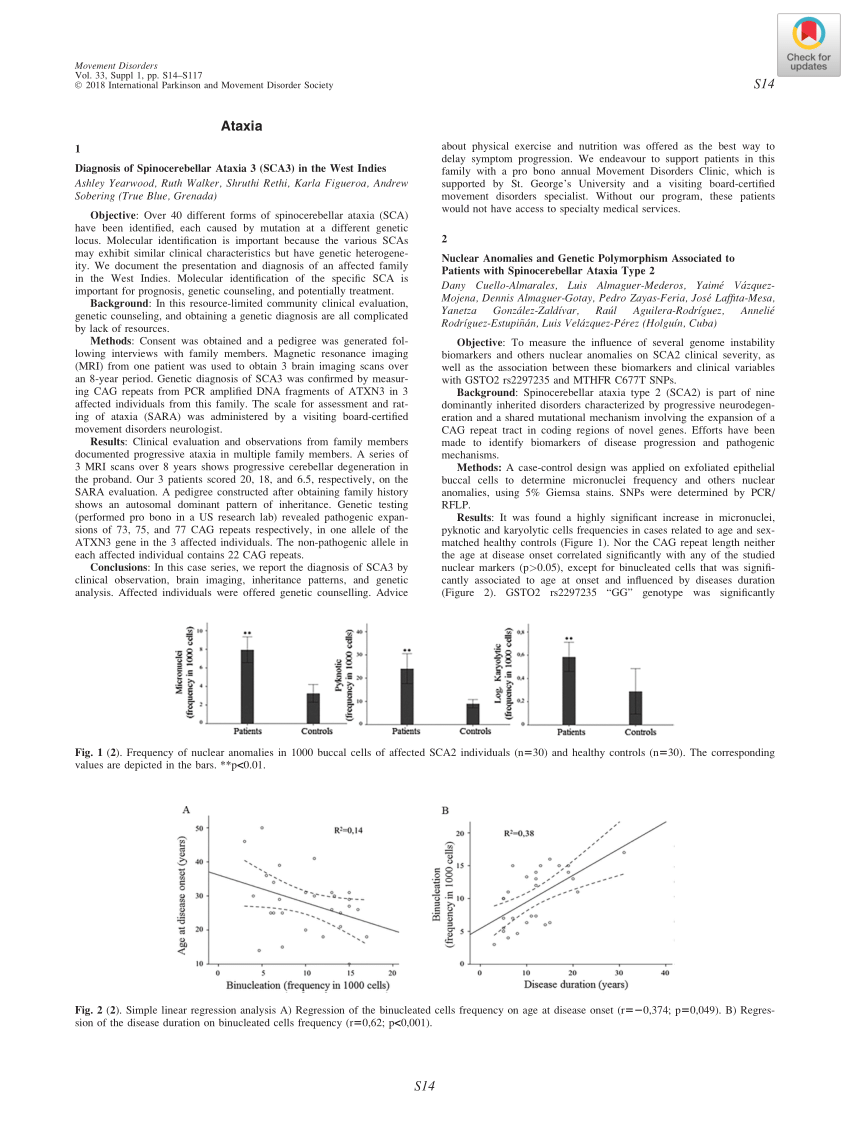
Inilah Daftar Obat Baru di Bulan April 2017 yang Disetujui FDA 4. Ingrezza. FDA menyetujui valbenazine dari Neurocrine Biosciences (Ingrezza) pada tanggal 11 April 2017. Ingrezza adalah obat pertama yang menerima persetujuan FDA untuk pengobatan tardive dyskinesia, sebuah kondisi neurologis yang ditandai dengan gerakan tak sadar berulang-ulang.

Ingrezza isi
Ingrezza® - ASD Healthcare You should use your clinical judgment to determine which product may be appropriate in each instance. Please refer to the product's information page for details on approved indications. Please consult the product labeling and associated ISI for the most accurate and current information about these products, including approved indications. PDF Prescribing Information - AUSTEDO Titrate at weekly intervals by 6 mg per day based on reduction of chorea or tardive dyskinesia, and tolerability, up to a maximum recommended daily dosage of 48 mg (24 mg twice daily) (2.1) Administer total daily dosages of 12 mg or above in two divided doses (2.1) The 41% Man | Institutional Investor The company's drug Ingrezza was approved last year to treat a movement disorder "associated with the long-term use of drugs for schizophrenia." Neurocrine climbed more than 100 percent last ...
Ingrezza isi. Inilah Daftar 5 Obat Baru di Bulan Oktober 2017 Ingrezza FDA menyetujui kapsul 80 mg valzenazine dari Neurocrine Biosciences (Ingrezza) pada tanggal 5 Oktober 2017. Obat tersebut, penghambat VMAT2 selektif yang diminum sekali sehari, bekerja dengan mengurangi jumlah dopamin yang dilepaskan di bagian otak yang mengendalikan gerakan dan fungsi motorik, yang membantu mengatur sinyal saraf pada ... Neurocrine Biosciences Inc (NBIX) Q1 2019 Earnings Call Transcript - Yahoo! INGREZZA remains affordable for patients with three-quarters of patients paying $10 or less per month out of pocket and INGREZZA is covered for approximately 90% of planned lives. Evercore ISI Starts Adamas Pharmaceuticals (ADMS) at Outperform Evercore ISI initiates coverage on Adamas Pharmaceuticals (NASDAQ: ADMS) with a Outperform rating and a price target of $85.00. Analyst Josh Schimmer has "extremely high conviction" in the... Evercore ISI Stick to Their Buy Rating for Neurocrine - Investing.com Evercore ISI analyst Joshua Schimmer maintained a Buy rating on Neurocrine (NASDAQ: NBIX) on Wednesday, setting a price target of $130, which is approximately 39.48% above the present share price...
Snippet roundup: Firsts for Chinese CAR companies, setbacks for J&J and ... Xenazine is a benchmark owing to pricing considerations - Leerink analysts reckon on Austedo costing just $45,000 per patient per year versus Xenazine's $83,000 - and Austedo and Ingrezza's relative safety could be key. Teva aims to file this year, while Neurocrine has already done so. J&J's PD-1 asset comes out of stealth mode September 22, 2016 Treatment for Adults With Tardive Dyskinesia (TD) | INGREZZA ... Whether your uncontrollable body movements are mild, moderate, or severe, connecting with a psychiatry or neurology healthcare provider who has experience diagnosing and treating TD is important. Enter your ZIP code to find a specialist near you and ask about reducing TD movements with INGREZZA—the only once‑daily treatment for TD. a. Valbenazin (Ingrezza) - Banyere ỌGwụ - 2022 Ndakpọ ma ọ bụ isi ọwụwa nwere ike bute ọdịda, ihe ọghọm, ma ọ bụ mmerụ ahụ dị njọ. Kedu ọgwụ ndị ọzọ ga -emetụta Valbenazine (Ingrezza)? Ọgwụ ndị ọzọ nwere ike imetụta valbenazine, gụnyere ọgwụ ndenye ọgwụ na ọgwụ, vitamin, na ngwaahịa ahịhịa. Home | Otsuka US Otsuka is a healthcare company driven by our purpose and defined by our beliefs. August 10, 2022.
Stocks To Watch: Top Biotech Scores Price Target Hikes As It ... - IBD The biotech's Ingrezza tardive dyskinesia treatment brought in $231.1 million in revenue, a 69.4% year-over-year jump. Tardive dyskinesia, an involuntary movement disorder and side effect of some... Stocks To Watch: Top Biotech Scores Price Target Hikes As It Nears Buy ... The biotech's Ingrezza tardive dyskinesia treatment brought in $231.1 million in revenue, a 69.4% year-over-year jump. Tardive dyskinesia, an involuntary movement disorder and side effect of some... Schizophrenia Treatment & Bipolar I Maintenance | ABILIFY MAINTENA ... ISI Block Title WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS. Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at increased risk of death (1.6 to 1.7 times) compared to placebo-treated patients. ABILIFY MAINTENA is not approved for the treatment of patients with dementia ... Neurocrine Biosciences [NBIX] Stock - Buy or Sell? | The Analyst San diego , may 23, 2022 /prnewswire/ -- neurocrine biosciences, inc. (nasdaq: nbix) today announced that it will present data on the use of ingrezza® (valbenazine) capsules for the treatment of tardive dyskinesia (td) at the american psychiatric association (apa) annual meeting being held in person may 21-25 in new orleans and virtually june ...
Neurocrine Biosciences Inc (NBIX) Q1 2021 Earnings Call Transcript Question from Josh Schimmer from Evercore ISI. Please go ahead. ... Since you grew INGREZZA, pretty rapidly to $1 billion -- nearly $1 billion in annual revenues last year, just on TD and just in ...
Neurocrine Biosciences, Inc. (NBIX) CEO Kevin Gorman on Q1 2022 Results ... From a regulatory perspective, INGREZZA was designated a breakthrough medicine by the FDA. It's also been a breakthrough in the sense of completely changing the paradigm of TD treatment. While as...
Important Safety Information | INBRACE Support Program | INGREZZA ... The most common side effect of INGREZZA is sleepiness (somnolence). Other side effects include changes in balance (balance problems, dizziness) or an increased risk of falls, headache, feelings of restlessness, dry mouth, constipation, and blurred vision. These are not all of the possible side effects of INGREZZA.
AUSTEDO® (deutetrabenazine) tablets | Healthcare Professionals Website Balance the risks of depression and suicidality with the clinical need for treatment of chorea. Closely monitor patients for the emergence or worsening of depression, suicidality, or unusual changes in behavior.
Approval For Neurocrine's Ingrezza Was No Surprise, But Lack ... - Yahoo! Shares of Neurocrine are up more than 23 percent on Wednesday. Ingrezza will be available at specialty pharmacies within a week's time, but Skorney says the company is expected to launch a...
Important Safety Information | INBRACE Support Program | INGREZZA ... INGREZZA is contraindicated in patients with a history of hypersensitivity to valbenazine or any components of INGREZZA. Rash, urticaria, and reactions consistent with angioedema (e.g., swelling of the face, lips, and mouth) have been reported. WARNINGS & PRECAUTIONS Somnolence INGREZZA can cause somnolence.
PDF 25 Words or Less 25 Words or Less Double Strip 25 Words or Less 25 Words or Less Double Strip Episode Name: Episode Number: 25W03018 Air Date: Duration: 7/13/2022 HD-25W03018 Rating: 0:30:01;00 TVPG
The Huntington's pipeline takes a blow | Evaluate One phase III candidate, Neurocrine's tardive dyskinesia drug Ingrezza is only being trialled for the chorea associated with the disease, and would not represent a disease-modifying therapy. At least one other mid-stage project is on shaky ground: Vaccinex's pepinemab failed its phase II study, Signal.
The Assessment and Treatment of Antipsychotic-Induced Akathisia The treatment of antipsychotic-induced akathisia should be personalised, with consideration of antipsychotic dose reduction, cessation of antipsychotic polypharmacy, and switching to an antipsychotic with a perceived lower liability for akathisia, before the use of adjuvant medications.
Neurocrine Biosciences (NBIX) Q1 2022 Earnings Call Transcript From a regulatory perspective, Ingrezza was designated a breakthrough medicine by the FDA. It's also been a breakthrough in the sense of completely changing the paradigm of TD treatment. What was...
Valbenazin (Ingrezza) - E Uiga I Vailaau - 2022 - medicalcareerinstitute O a isi vailaʻau o le a aʻafia ai Valbenazine (Ingrezza)? O isi vailaʻau e mafai ona aʻafia ai le valbenazine, e aofia ai fualaʻau ma vailaʻau oona, vailaʻau, ma vailaʻau. Taʻu i lau fomaʻi au vailaʻau uma o loʻo iai nei ma isi vailaʻau o loʻo e inuina pe a amata pe le toe faʻaaogaina. O fea ou te maua ai nisi faʻamatalaga ...
INQOVI® (decitabine and cedazuridine) tablets | MDS Treatment The most common side effects of INQOVI include: low white blood cell count (leukopenia), low platelets in your blood (thrombocytopenia), low white blood cell count (neutropenia), low red blood cell count (anemia), tiredness, constipation, bleeding, muscle pain, pain or sores in your mouth or throat, joint pain, nausea, shortness of breath ...
The 41% Man | Institutional Investor The company's drug Ingrezza was approved last year to treat a movement disorder "associated with the long-term use of drugs for schizophrenia." Neurocrine climbed more than 100 percent last ...
PDF Prescribing Information - AUSTEDO Titrate at weekly intervals by 6 mg per day based on reduction of chorea or tardive dyskinesia, and tolerability, up to a maximum recommended daily dosage of 48 mg (24 mg twice daily) (2.1) Administer total daily dosages of 12 mg or above in two divided doses (2.1)
Ingrezza® - ASD Healthcare You should use your clinical judgment to determine which product may be appropriate in each instance. Please refer to the product's information page for details on approved indications. Please consult the product labeling and associated ISI for the most accurate and current information about these products, including approved indications.







:max_bytes(150000):strip_icc()/GettyImages-171296967-3383cfb26dd346d894a5c41e6ac722af.jpg)









Post a Comment for "43 ingrezza isi"