45 lewis symbols
4.4 Lewis Symbols and Structures - Chemistry: Atoms First 2e - OpenStax We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 4.10 shows the Lewis symbols for the elements of the third period of the periodic table. Lewis Symbols and Structures | Chemistry for Majors: Atoms First ... A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 1. Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table.
Lewis Structures: Dot Symbols, Diagram, Examples - Embibe The Lewis structure was named after Gilbert N. Lewis, who introduced it in his \(1916\) article "The Atom and the Molecule." Learn 11th CBSE Exam Concepts. In a Lewis Structure, electrons are represented as "dots" surrounding the central metal atom. The central metal is denoted by using its chemical symbol from the Periodic Table.
Lewis symbols
Lewis Symbols and Structures | Chemistry I | | Course Hero We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 1. Lewis symbols illustrating the number of ... Lewis Symbols and Structures - Chemistry We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: [link] shows the Lewis symbols for the elements of the third period of the periodic table. Lewis symbols illustrating the number of valence ... 4.4 Lewis Symbols and Structures - General Chemistry 1 & 2 Lewis Symbols. We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 1. Lewis symbols illustrating the ...
Lewis symbols. Lewis Dot Symbols and Lewis Structures | Boundless Chemistry | | Course ... The Lewis symbol for carbon: Each of the four valence electrons is represented as a dot. Electrons that are not in the valence level are not shown in the Lewis symbol. The reason for this is that the chemical reactivity of an atom of the element is solely determined by the number of its valence electrons, and not its inner electrons. Lewis ... Lewis Symbols and Structures | Chemistry for Majors A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 1. Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table. 6.01 Lewis symbols - Week 6 | Coursera These structures provide information about the types of bonds (single, double, or triple) as well as the connectivity of atoms. By knowing the Lewis structure, we can also predict the three-dimensional geometry of an individual molecule. 6.01 Lewis symbols 4:36. 6.02 Covalent bonds 5:19. 6.03 Electronegativity 11:21. Lewis Symbols or Diagrams - Elmhurst University A Lewis Symbol consists of the element symbol surrounded by "dots" to represent the number of electrons in the outer energy level as represented by a Bohr Diagram. The number of electrons in the outer energy level is correlated by simply reading the Group number. Lewis symbols for oxygen, fluorine, and sodium are given in the diagram on the left.
Lewis Symbols and Structures (4.4) - Chemistry 110 - Unizin We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol. consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 4.9 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 4.9 Lewis symbols illustrating the number of ... Lewis Symbols and the Octet Rule | Chemistry | JoVE The Lewis symbol for neon has eight dots, two dots on each side, representing the filled electron configuration; in other words, an octet. The octet rule states that an atom tends to lose, gain, or share electrons in the form of bonds until a stable electron configuration, an octet, is reached. Consider carbon dioxide. 7.3 Lewis Symbols and Structures - Chemistry A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 1. Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table. Lewis symbols can ... Lewis Symbols and Structures - Introductory Chemistry Lewis Symbols. We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: The table below shows the Lewis symbols for the elements of the third period of the periodic table. Lewis symbols illustrating the ...
Lewis Symbols and Structures - chem-textbook A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: shows the Lewis symbols for the elements of the third period of the periodic table. Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table. Lewis symbols can also be used to ... Lewis Symbols Flashcards | Quizlet What are lewis symbols? the dots representing the valance electrons in the atom. how do we write them? 1. Determine # of valence electrons 2. Put that number of dots around elemental symbol. since electrons have a 1- charge, they want to be_____ from each other. far away. The Lewis - A symbol in Freemasonry. A Lewis is an operative tool, Masonic symbol, or young man adopted by a Lodge. A lewis is an instrument in operative masonry. It is a cramp iron which is inserted into a cavity prepared for that purpose in any large stone, so as to give attachment to a pulley and hook whereby the stone may be conveniently raised to any height and deposited in ... Michael Schumacher | Formula 1 Wiki | Fandom Formula One Career [] 1991 [] Main article: 1991 Michael Schumacher Season Michael Schumacher began his career in the relatively uncompetitive debutant Jordan car, replacing jailed Bertrand Gachot at the Belgian Grand Prix.Surprisingly, he qualified 7th, with only the McLaren-Honda, Ferraris, a Williams-Renault and a Benetton-Ford in front. His clutch failed on the way up to Eau Rouge, but his ...
Lewis Symbols - Chemical Bonding and Molecular Structure Lewis Symbols. A group of atoms existing together as one species and having characteristic properties is called a molecule. This force which holds the atoms together within a molecule is called a covalent bond. Noble gases neither combine chemically with any other element nor among themselves i.e. they are chemically inert.
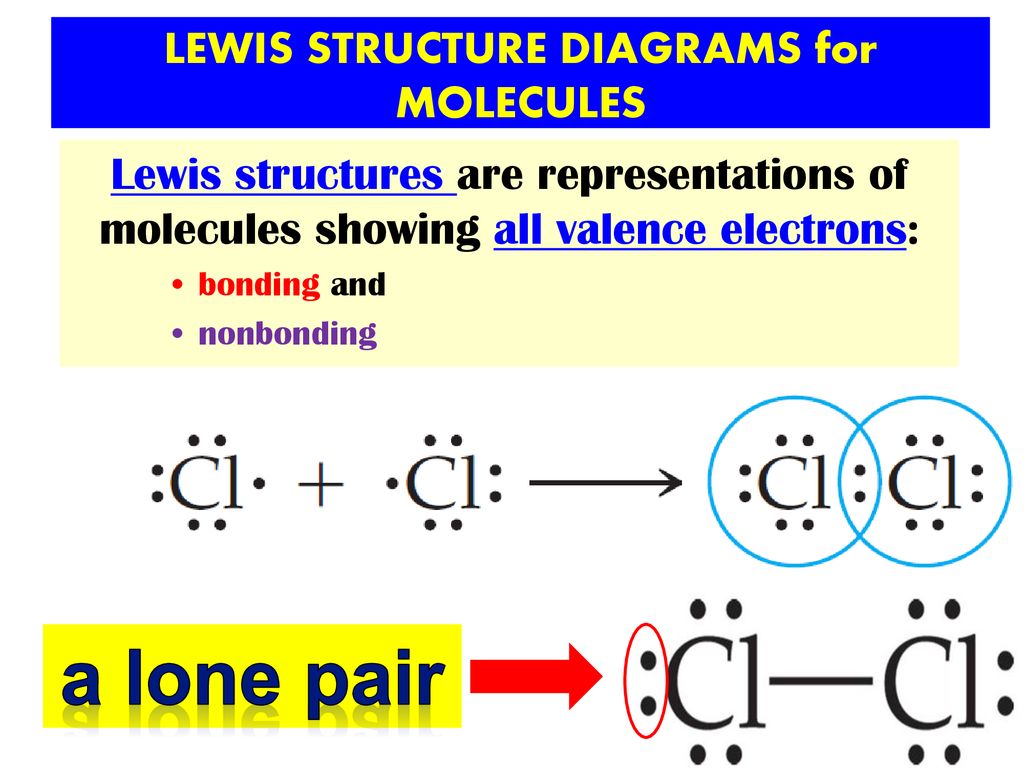
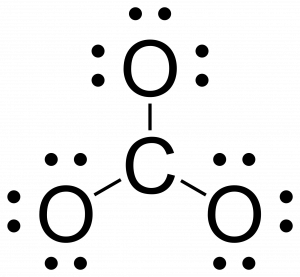
Lewis structure - Wikipedia Lewis structures, also known as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures ... Lewis structures show each atom and its position in the structure of the molecule using its chemical symbol. Lines are drawn between atoms that are bonded to one another (pairs of dots can be used instead of ...
6.1 Lewis Symbols and Structures - Chemistry Fundamentals We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 6.1.1 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 6.1.1 Lewis symbols illustrating the number ...
7.3 Lewis Symbols and Structures - Chemistry - Pressbooks Lewis Symbols. We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 1. Lewis symbols illustrating the ...
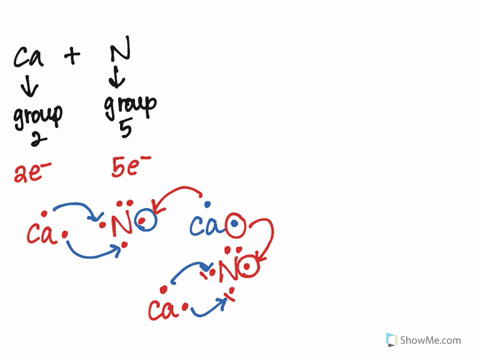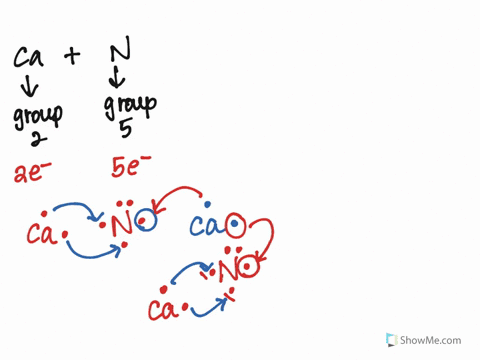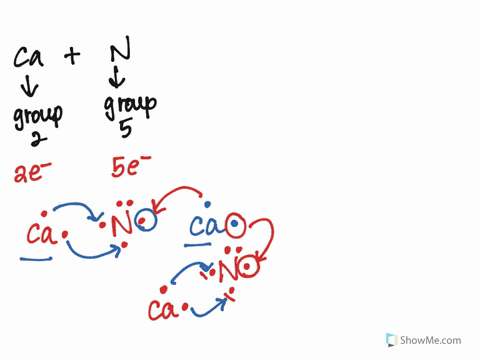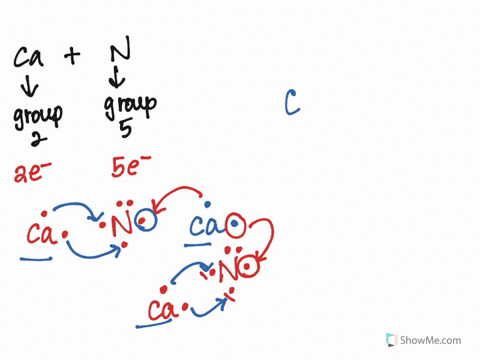
Solved 51. Give the correct Lewis symbols for the 2nd period | Chegg.com Give the correct Lewis symbols for the 2nd period elements. 52. A double covalent bond involves the sharing of atoms. - 53. Which of the following elements typically cannot support a triple bond? a. O d. N b. C e. a and b c. H f. a and c 57. The molecule ammonia, NH, has 54. The bond strength of an ionic bond depends on the of the ions.
Lewis Structure | Encyclopedia.com Lewis symbols are a simple way of visualizing the valence electrons in an atom. In a Lewis symbol, the symbol for the element is used to represent the atom and its core electrons. Dots placed around the atom are used to indicate the valence electrons. When combined to form Lewis structures, Lewis symbols make it possible to predict the shape of ...
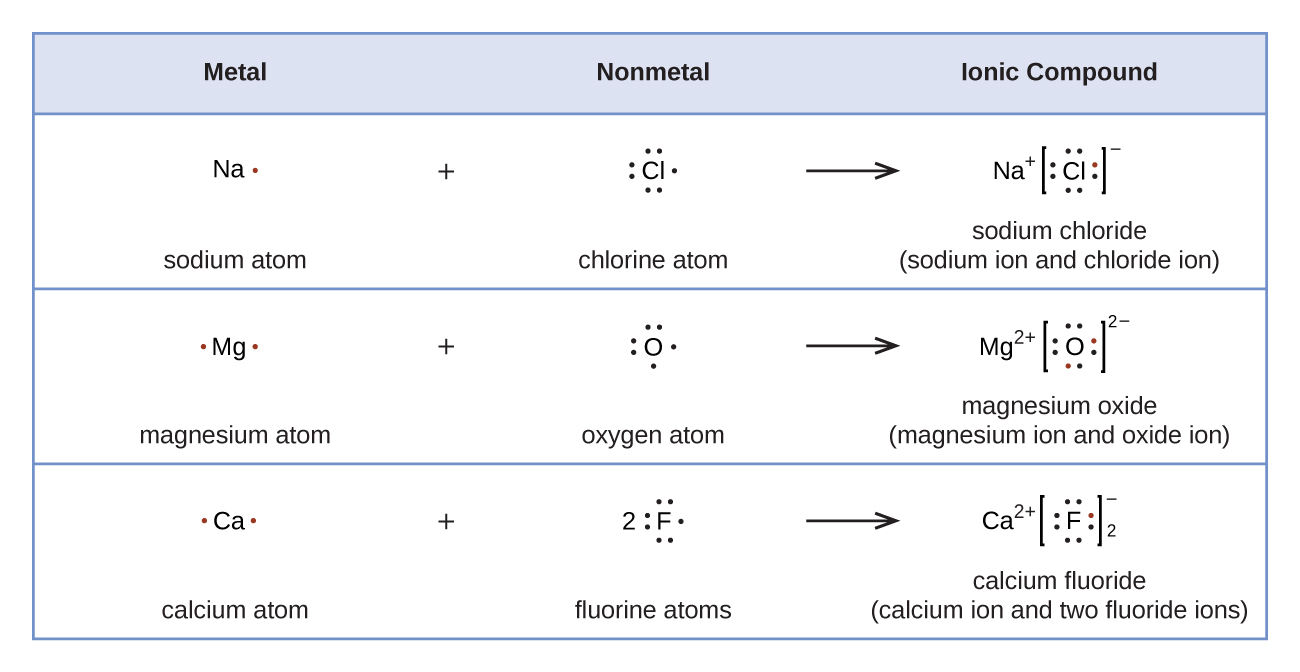
Lewis Symbols | Chemical Bonding - Nigerian Scholars Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table. Lewis symbols can also be used to illustrate the formation of cations from atoms, as shown here for sodium and calcium: Likewise, they can be used to show the formation of anions from atoms, as shown here for chlorine and ...
Lewis Symbols and Structures - Chemistry 2e We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: (Figure) shows the Lewis symbols for the elements of the third period of the periodic table. Lewis symbols illustrating the number of valence ...
lewis symbol - Copy And Paste You can use lewis symbol to copy and paste it in. Facebook; Instagram; Whatsapp; Twitter; Pinterest; Tumblr; TikTok; Meaning of lewis symbol. The use of lewis symbol can have different meanings. About unicode lewis symbol. Unicode is a system of programming symbols used by programming equipment for the storage and forwarding of data in format ...
Lewis Symbols - Chemistry LibreTexts A Lewis Symbol is constructed by placing dots representing electrons in the outer energy around the symbol for the element. For many common elements, the number of dots corresponds to the element's group number. Below are Lewis Symbols for various elements. Notice the correspondence to each element's group number.
4.4 Lewis Symbols and Structures - General Chemistry 1 & 2 Lewis Symbols. We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 1. Lewis symbols illustrating the ...
Lewis Symbols and Structures - Chemistry We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: [link] shows the Lewis symbols for the elements of the third period of the periodic table. Lewis symbols illustrating the number of valence ...
Lewis Symbols and Structures | Chemistry I | | Course Hero We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 1. Lewis symbols illustrating the number of ...




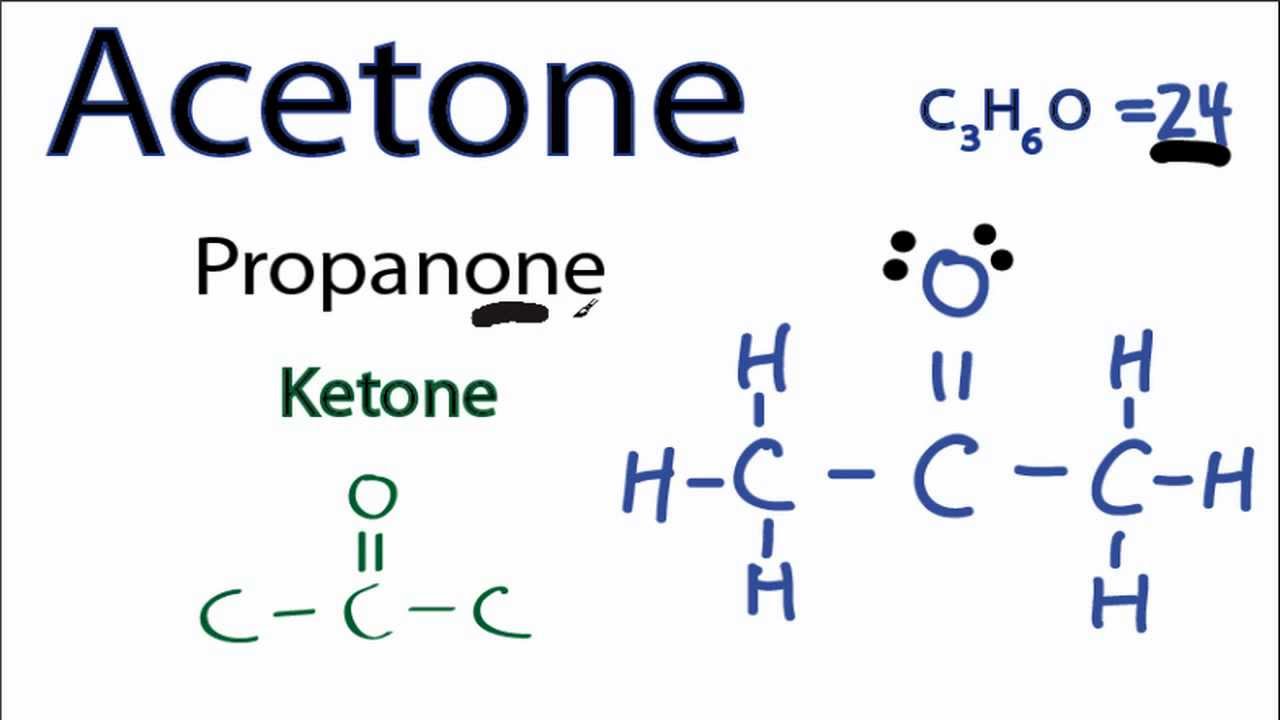
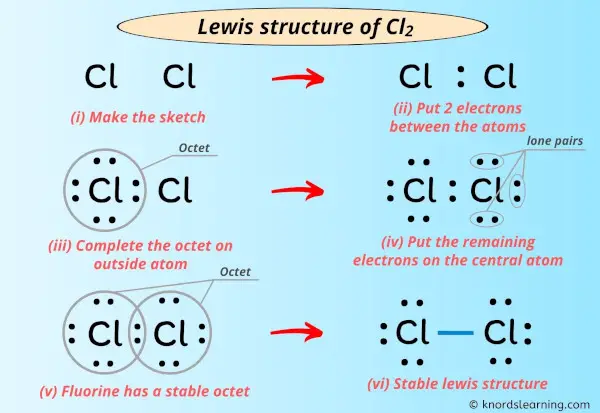

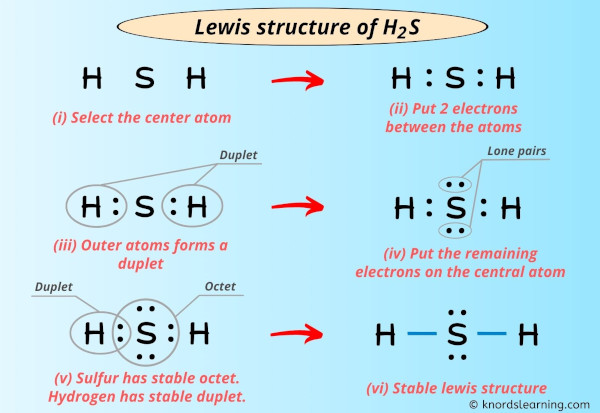






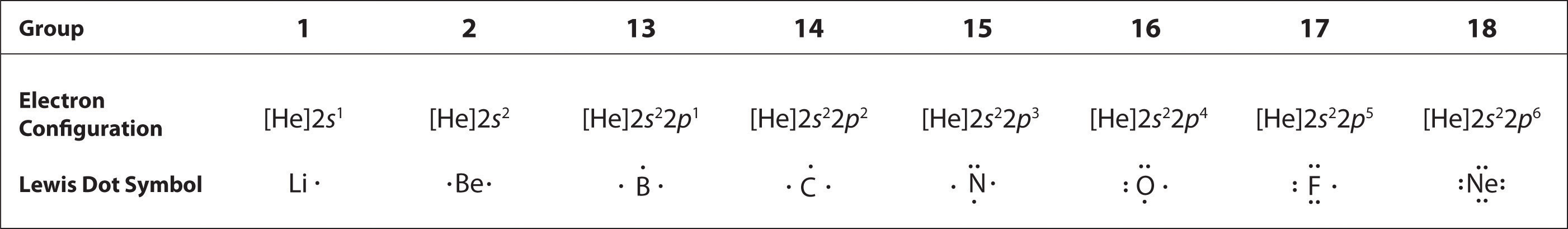

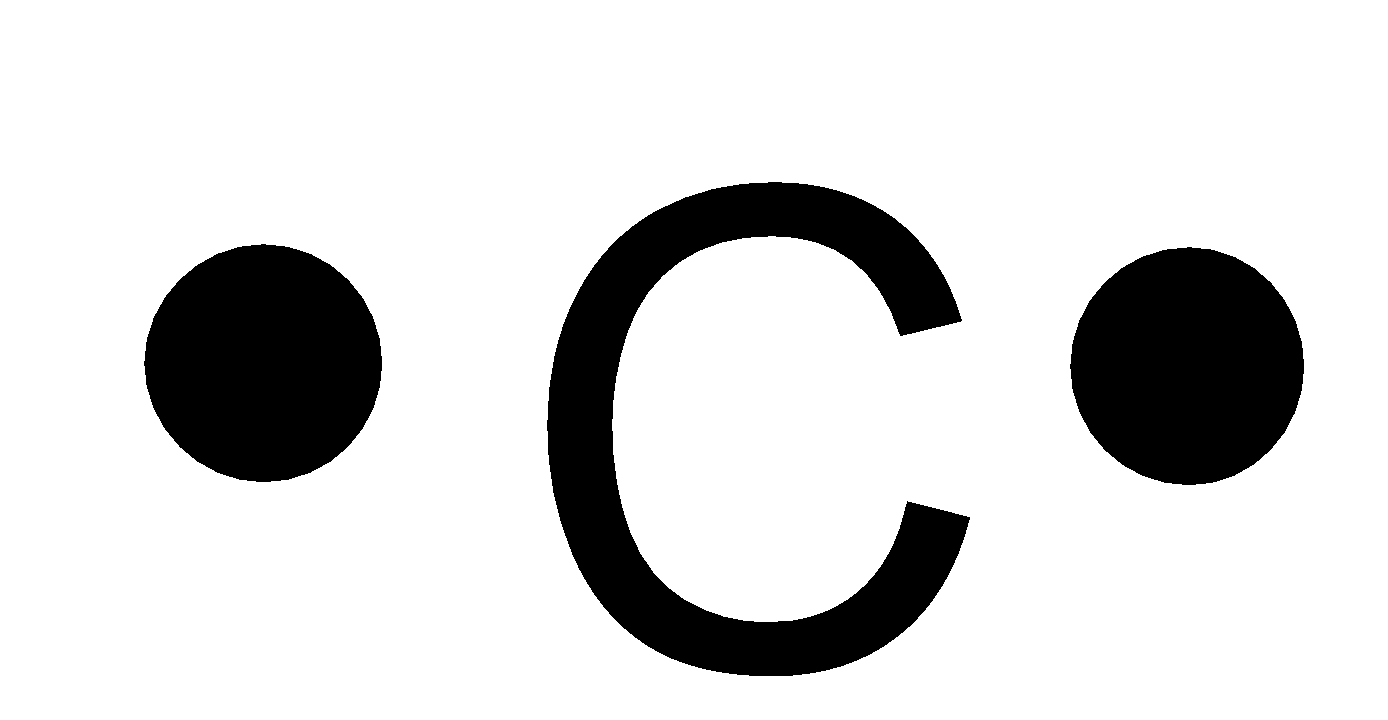
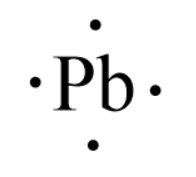










/ScreenShot2018-11-19at11.40.52PM-5bf3909a46e0fb00510dbd6d.png)


Post a Comment for "45 lewis symbols"